|
Key Takeaways
|
Many aspects of clinical trials, such as managing patient enrollment, drug supply, and randomization, can be challenging. A true companion, “Interactive Response Technology” for clinical trials addresses these challenges with high efficiency.
The market value of IRT clinical trials is valued at roughly USD 16.7 billion in 2024 and is projected to grow with a CAGR of 21.6% from 2025 to 2033, reaching around USD 91.4 billion by 2033.
A central question: What is interactive response technology? And why is this tech integral to clinical trials?
Let’s get an insightful overview of IRT, understanding what it is and its impact on clinical trial operations.
Interactive Response Technology (IRT): In Detail
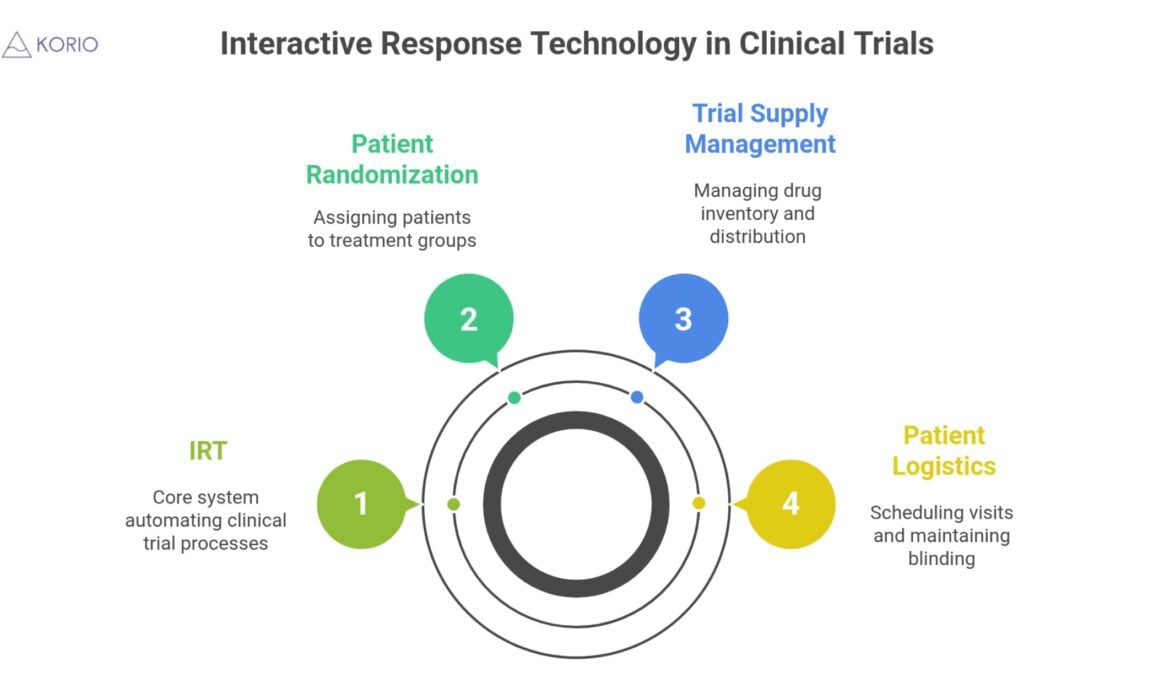
IRT is a software system designed to streamline and automate critical processes within clinical trials.
Most common usage includes:
01. Patient Randomization: Patients are assigned to different arms in a trial.
02. Trial Supply Management: Dispensation, inventory, and replenishment of drugs to clinical trial sites.
03. Patient Logistics: Scheduling of patients and ensuring that patients remain blinded on trial.
| NOTE: IRT enables real-time management via a web-based interface, eliminating errors commonly made manually and increasing efficiency at all points of a trial. |
The Reasons for IRT Usage
Many students interested in understanding these software systems wonder why IRT is used:
| S.No | Use Case | Description (Reason for usage) |
| 01. | Patient Randomization | Ensures fair and unbiased assignment to treatment arms following trial protocols |
| 02. | Trial Supply Management | Oversees drug inventory, distribution, and reordering, avoiding stockouts and waste |
| 03. | Visit Scheduling & Compliance | Tracks patient visits and adherence to schedules, improving trial data quality |
| 04. | Regulatory Compliance | Maintains audit trails and data integrity, ensuring compliance with standards such as Good Clinical Practice (GCP) and FDA guidance |
Table: Why IRT is used for certain use cases of clinical trials
Understanding IRT in Clinical Trials
Within clinical trials, this technology allows a clinical team to:
A. Randomize patients between lines to end up with a perfect randomization to meet the protocol requirements and achieve an unbiased manner of treatment allocation.
B. Automate assignment of drugs and monitoring drug inventory in order to avoid overstocking or running out of drugs at trial sites.
C. Streamline visit schedules and patients and monitor compliance and data integrity.
D. Trial blinding by limiting the access of patients to their treatment by the staff.
IRT is used in studies ranging all the way through Phase I through Phase IV and has been highly advantageous in complex trials and/or trials with multiple sites, in therapeutic areas such as oncology.
Clinical trial teams can automate these procedures, minimize manual efforts, eliminate risks, and speed up study timelines.
How Does IRT Work?
Here are the steps of how IRT functions:
-
- Data Integration: IRT can collect and process patient data and supply real-time information.
-
- Algorithms of Randomization: Patients are randomly sampled using predetermined schedules; this is done to reduce bias.
-
- Supply Chain Coordination: The system predicts the drug usage, monitors stock levels, and initiates refills.
-
- Access and Communication: The interfaces enable sites and sponsors to have immediate access to crucial trial data in order to make the right decisions.
-
- Regulatory Compliance: Auto-audit trails will ensure every workflow is capable of compliance with regulations.
What Makes IRT Apart From Similar System Software
Many benefits of IRT make it a must for clinical trials. These are:
-
- Accuracy: Alongside real-time data access and automation, the technology gives high-accuracy results at certain phases of the trial because of the reduction of human errors.
-
- Scalability: The suitable use from small Phase 1 trials as well as for large, multi-site Phase 3/4 studies.
-
- Flexibility: Web-based interfaces accommodate diverse site capabilities and user preferences.
-
- Compliance Support: Audit trails and documentation improve regulatory audit readiness.
Perfect Alignment: Korio and IRT Principles
Korio is your cutting-edge Randomization and Trial Supply Management (RTSM) software platform, designed specifically to tackle the challenges of clinical trials and prevent them in the first place.
With flexibility, quality, and speed, it allows sponsors to efficiently manage patient randomization, drug supply logistics, and site operations across complex and global studies.
With over 20+ years of RTSM hands-on experience, Korio supports early-phase to large Phase 3 trials and has features such as continuous audit trails, automated testing, and integration-ready architecture, ensuring high data integrity and compliance throughout the trial lifecycle.
Final Thoughts
IRT is a pillar of present-day clinical trials. It automates the process of randomizing the patients, addressing logistical challenges. By decreasing human intervention and error, IRT helps the clinical research staff devote their time and energy to providing high-quality results within a shorter amount of time.
In international trials, IRT is essential in accounting for the multifaceted nature with regard to geography, regulatory guidelines, and the diverse population. There is, consequently, the need to understand and embrace IRT in the current clinical trial environment.
Frequently Asked Questions ( FAQs)
01. What are the major uses of interactive response technology?
It can be applied to randomize patients, control drug supplies, schedule visits, and control trial compliance.
02. What is an IRT system in a clinical trial?
An IRT system is a system software that provides automation of vital duties in the running of a trial, such as supply chain management and randomization.
03. How is IRT an improvement in the line of clinical trials?
IRT increases efficiency, minimizes manual errors, empowers access to real-time data, and ensures legislative compliance.
04. Is IRT used in all phases of clinical trials?
Yes, IRT helps address different trial operational requirements during the Phase I to Phase IV.
05. What are other names of IRT?
Other terms include Randomization and Trial Supply Management (RTSM), Interactive Voice Response System ( IVRS ), and Interactive Web Response System (IWRS).